Fist law of thermodynamics

First Law of Thermodynamics

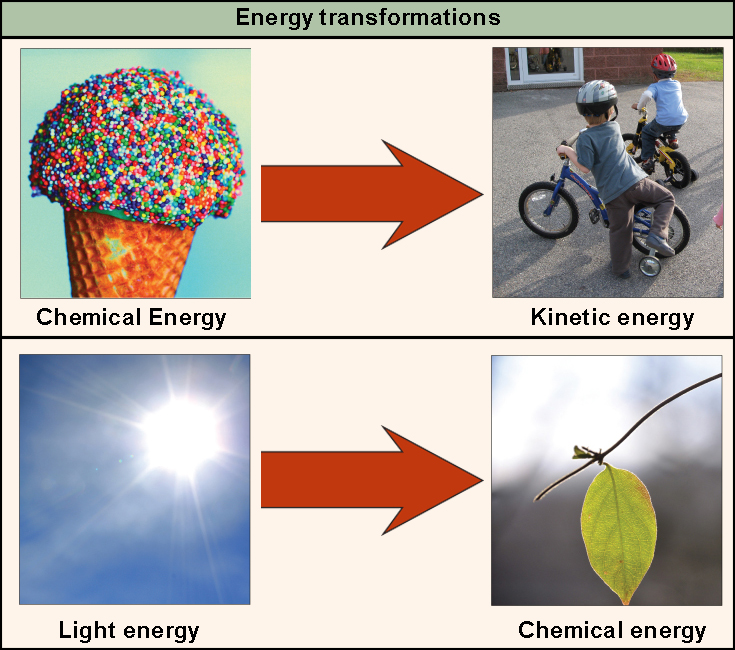



The first law of thermodynamics is a version of the law of conservation of energy, adapted for thermodynamic systems. The law of conservation of energy states.


The first law of thermodynamics is the application of the conservation of The first law makes use of the key concepts of internal energy, heat, and system work.

Navigation menu

I Becky Age: 26. I consider myself to be the perfect playmate from my classy, girly next door bombshell look to my unforgettable expertise between the sheetsMy name is MayaWaiting for you
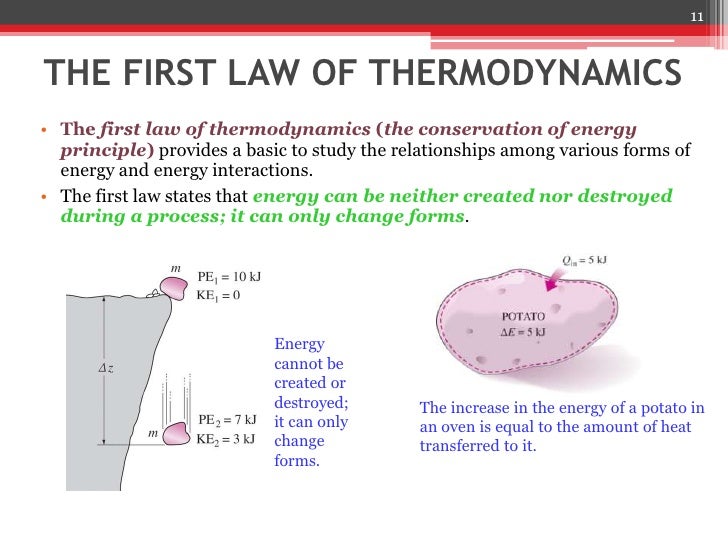
What Is the First Law of Thermodynamics?

Chloe Age: 22. Ciao ,minimum respectpolite and sophisticated lady in public, submissive slut in the bedroom.


The first law of thermodynamics applies the conservation of energy principle to systems where heat transfer and doing work are the methods of transferring.

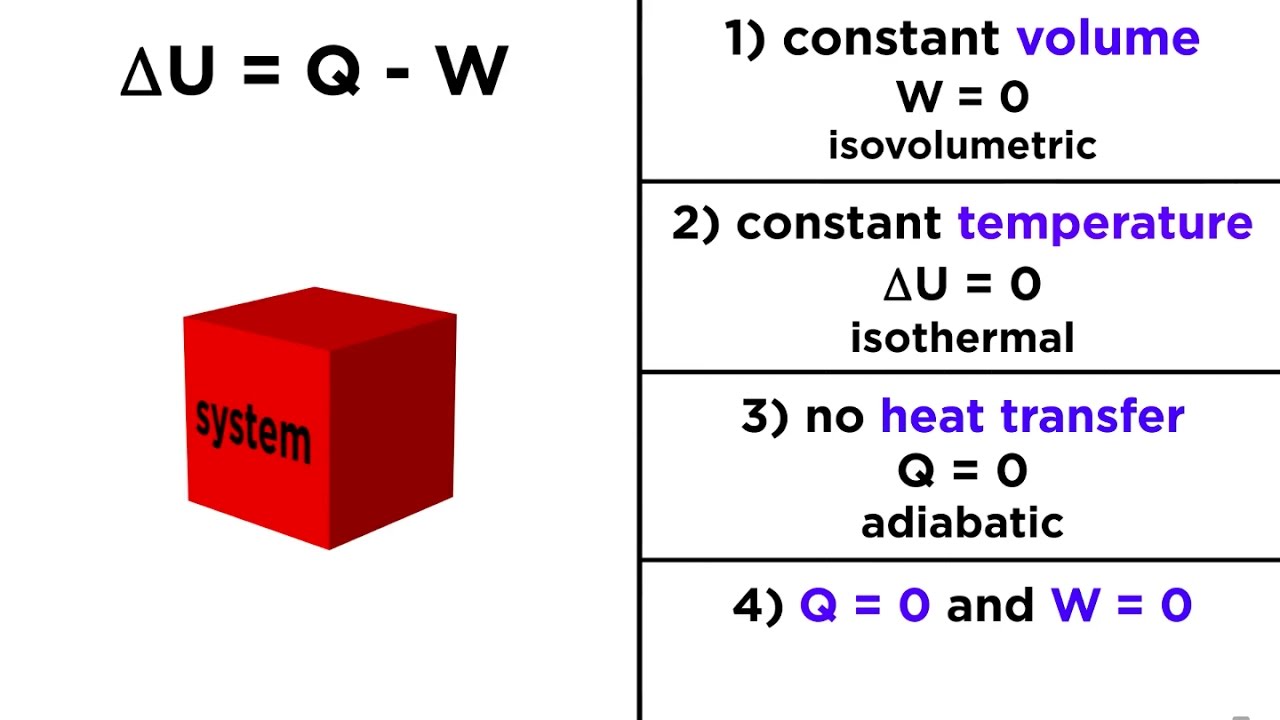

Description:What Is the First Law of Thermodynamics? The movement can be harnessed to do work equal to the total force applied to the top of the piston times the distance that the piston moves. GoodIll Shutterstock The First Law of Thermodynamics states that heat is a form of energy, and thermodynamic processes are therefore subject to the principle of conservation of energy. This means that heat energy cannot be created or destroyed. It can, however, be transferred from one location to another and converted to and from other forms of energy.
















































User Comments 5
Post a comment
Comment: